Back to: CHEMISTRY SS1
Welcome to class!
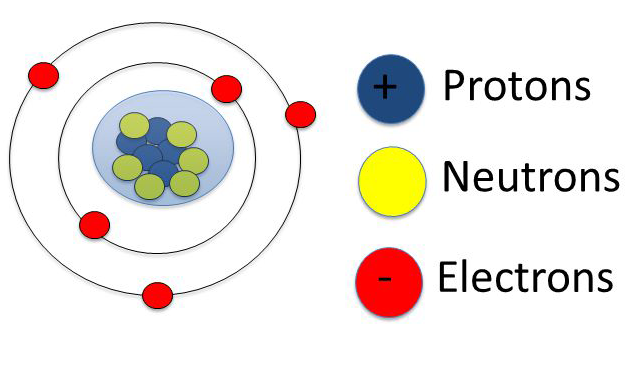
In today’s class, we will be talking about the structure of the atom. Enjoy the class!
STRUCTURE OF THE ATOM: ORBITALS AND ELECTRONIC STRUCTURE OF THE ATOM
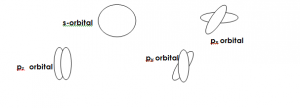
ORBITALS OF THE ATOM
The region in space within which there is a maximum possibility of finding an electron in an atom is called an ATOMIC ORBITAL. We have s, p, d, and f orbitals.
Shapes of s and p orbitals are as follows:
EVALUATION
- Define atomic orbital
- State the shape of (i) S-orbital (ii) P-orbital.
ELECTRONIC STRUCTURE OF THE ATOM
With the knowledge of atomic orbital the spectra of more complex atoms than hydrogen atom can be explained as follows:
- (a) that within a given principal quantum number or energy level, there are sub energy levels, i.e. energy levels otherwise called K, L, M, N, O, P AND Q shells have sub-energy levels otherwise called s, p, d and f orbitals
(b) The total number of sub-shells within a shell is given by n2
while the total maximum number of electrons is given by 2n2 where n is the number of energy levels.
Energy Level Number of orbitals Maximum No of electrons
n = 1(K- shell) 12 =1 2´12 =2
n =2 (L- shell) 22 = 4 2´22 = 8
n = 3 (M- shell) 32 = 9 2´32 = 18
n = 4 (N- shell) 42 =16 2´ 42 = 32
n = 5 (O- shell) 52 = 25 2´ 52 =50
n = 6 (P-shell) 62 = 36 2´62 =72
n = 7 (Q – shell) 72 = 49 2´72 = 98
(c) In a given orbital there could be a maximum of only two electrons and electron in all orbitals of the same type within a principal quantum number possess equal energies.
(d) The electrons in the different subshells or orbitals within a principal quantum number do not all have equal energies.
The gradation of energies of orbitals is as follows:
1s< 2s<2p<3s<3p<3d<4s<4p<4d—
EVALUATION
- State the first five energy levels.
- Calculate the total no of electrons when n is equal to (i) 2 (ii) 4.
FILLING OF ELECTRONS IN ORBITALS
In filling electrons into the atoms of elements, considerations are given to the conditions laid down by Aufbau Principle, Pauli exclusion principle and Hund’s rule of maximum multiplicity.
- AUFBAU PRINCIPLE: states that electrons go in to fill orbitals of lower energy first before filling orbitals of higher energy and each orbital may hold up to two electrons.
- PAULI EXCLUSION PRINCIPLE: states that no two electrons have identical sets of the four quantum numbers {n, l, m and s in an atom}.
- HUND’S RULE OF MAXIMUM MULTIPLICITY: state that in filling degenerate orbitals with electrons, electrons go in singly first before pairing up occurs.
EVALUATION
State the following principle
{a} Aufbau Principle {b} Hund’s rule.
QUANTUM NUMBERS
Studies show that the energy of an electron may be characterized by four quantum numbers. These are
{1} The principal quantum number represented by n with integral values of 1,2,3,4 e.t.c.
{2} The subsidiary or Azimuthal quantum number represented by l with integral values ranging from O to (n-1).
{3} The magnetic quantum number represented by m with integral values ranging from –l to +l.
{4} The spin quantum number represented by s with integral values – 1/2 and = 1/2.
Element Atomic Number Electronic configuration.
- H 1 1S1
- He 2 1S2
- Li 3 1S2 2S1
- Be 4 1S2 2S2
- B 5 1S2 2S2 2P1
- C 6 1S2 2S2 2P2
- N 7 1S2 2S2 2P3
- O 8 1S2 2S2 2P4
- F 9 1S2 2S2 2P5
- Ne 10 1S2 2S2 2P6
- Na 11 1S2 2S2 2P6 3S1
- Mg 12 1S2 2S2 2P6 3S2
- Al 13 1S2 2S2 2P6 3S2 3P1
- Si 14 1S2 2S2 2P6 3S2 3P2
- P 15 1S2 2S2 2P63S233P3
- S 16 1S2 2S2 2P63S2 3P4
- Cl 17 1S2 2S2 2P6 3S2 3P5
- Ar 18 1S2 2S2 2P6 3S23P6
- K 19 1S2 2S2 2P6 3S23P6 4S1
- Ca 20 1S2 2S2 2P6 3S23P6 4S2
GENERAL EVALUATION
- State the four quantum numbers.
- Write the electronic configuration of the following a) Mg b) S2- c) Ca2+ d) Si
READING ASSIGNMENT
- New School Chemistry By O.Y. Ababio pg 49-54.
WEEKEND ASSIGNMENT
- Which of the following orbitals is spherical in shape? (a) s (b) p (c) d (d) f
- Which of the following shells have a maximum of eight electrons? (a) k (b) l (c) m (d) n
- 1s2 2s2 2p6 3p1 is the electronic configuration of (a) potassium (b) calcium (c) sodium (d) aluminum.
- “No two electrons have identical sets of four quantum numbers”. This statement is (a) Aufbau principle (b) Pauli exclusion (c) Hund’s rule (d) Rutherford’s model.
- Which of the quantum number is represented by L? (a) principal quantum no (b) subsidiary quantum no (c) magnetic quantum (d) spin quantum.
THEORY
- State the following principle (a) Pauli Exclusion Principle. (b) Aufbau principle.
- Write the electronic configuration of (a) Oxygen (b) Calcium (c) Chloride ion (Cl–) (d) Aluminum ion (Al3+)
We hope you enjoyed the class.
Should you have any further question, feel free to ask in the comment section below and trust us to respond as soon as possible.
