Topic: ALKYNES
e.g. Ethyne
Alkynes are the homologous series of unsaturated hydrocarbon with a general molecular formula CnH2n-2.
Alkynes show a high degree of unsaturation than alkenes, hence, they are chemically more reactive than the corresponding alkenes or alkanes.
Examples are :
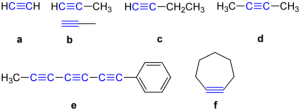
ETHYNE
Ethyne is the first member of the alkyne series. It has a molecular formula, C2H2, and a structural formula of HC = CH.
Laboratory Preparation
Ethyne is usually prepared in the laboratory by the action of cold water on calcium carbide. The reaction is carried out on a heap of sand to prevent the flask from cracking as a result of the large quantity of heat evolved.
Evaluation
- Write and name all possible structure of hexyne
- How can you prepare a few jars of ethyne in the laboratory?
NOMENCLATURE: The naming of alkyne are obtained by substituting “ane” in alkanes with ‘ene’.
Physical Properties
- Ethyne is a colourless gas with a characteristic sweet smell when pure.
- It is only sparingly soluble in water
- It is slightly less dense than air.
- It is unstable and may explode on compression to liquid.
Chemical Properties
- Combustion:- it undergoes combustion reaction in air to form water and carbon(iv) oxide
2C2H2 + 5O2 2H2O + 4CO2.
NB: In limited air, it burns with very smoky and luminous flame because of its high carbon content. But in plenty of air and appropriate proportion, it burns with non-luminous very hot flame of about 3000oC.
- Additional Reaction:-Ethyne undergoes addition reaction to produce unsaturated product with double bonds and then a saturated compound with single bond.
a. With hydrogen in the presence of nickel as catalyst.
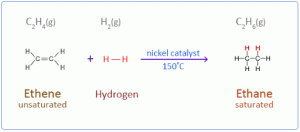
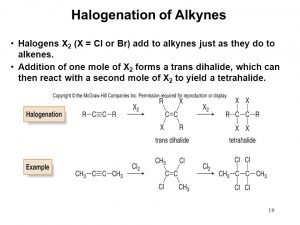
b. Halogenation: e.g Cl2, Br2, I2
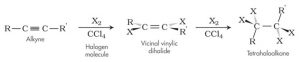
c. Addition of Halides;
Hydrogen halide reacts with ethyne to produce halo-alkene and further halogenation produce halo-alkane.
E.g.:
- Addition reaction with water through dilute tetraoxosulphate (vi) acid in the presence of CuSO4 as catalyst to form ethanol.
- Addition reaction with alkaline KMnO4 added to ethyne, it first turns to green from purple and then to colourless.
- Polymerization
In the presence of complex organic –nickel as catalyst to produce benzene.
3 C2H2 C6H6
3 (H – C = C – H ) C6H6
- Substitutional Reaction
- With ammomiacal solution of copper (1) chloride to form reddish brown copper (I) dicarbide
C2H2 + 2CuCl Cu2C2 + 2HCl
H – C = C – H + 2CuCl Cu – C = C- Cu + 2HCl
- With ammomiacal silver trioxonitrate (v) to form white silver dicarbide
C2H2 + 2AgNO3 Ag2C2 + 2HNO3.
H- C = C – H + 2AgNO3 Ag – C = C – Ag + 2HNO3.
N.B: Alkynes can be distinguished from alkene by reacting with ammonical metals of copper(I) chloride and silver trioxonitrate (vi).
USES
- It is used to produce oxyacetylene flame for cutting and welding of metals
- Used in the manufacture of PVC plastics
- It is used in miner’s lamp
- Used in making synthetic fibre
- It is also used in making artificial rubber
Test for Unsaturation
Unsaturated compound decolorizes bromine water.
Evaluation
- Give a chemical test to distinguish between alkyne and alkene.
- Describe a test for unsaturated compounds
READING ASSIGNMENT
New School Chemistry by Y. O Osei yaw Ababio Page
WEEKEND ASSIGNMENT
- The concentration of hydrogen ion in a neutral solution is
(a) 10-6 moldm-3 (b) 10-7moldm-3 (c) 5 x 10-7 moldm-3 (d) 1 x 10-8mol dm-3
- Hydrogen can be prepared in a large scale using the
(a) Harber’s process (b) Down’s process (c ) Bosh Process (d) Contact Process.
- Which of the following hydrocarbons is alkyne?
(a ) C2H4 (b) C2H6 (c) C2H2 (d) C3H8
- The final product of complete reaction between ethyne and hydrogen gas is (a) ethane (b)methyl ethane ( c) ethane ( d) hydroethyne
- Ethyne polymerizes in the presence of organonickel complex as catalyst to form (a) polythene (b) benzene (c) polythene (d) methyl benzene.
Theory
- a. Calculate the H+ of a solution whose PH is 5.
- State three (3) uses of ethyne
- a. With the aid of labeled diagram, describe the laboratory preparation of ethyne. b. Give a chemical test to distinguish between ethane and ethyne.
