Back to: CHEMISTRY SS2
Welcome to Class !!
We are eager to have you join us !!
In today’s Chemistry class, We will be learning about Compounds of Sulphur. We hope you enjoy the class!
CONTENT
- H2S, SO2 and SO3: Preparation, Properties and Uses
- Tetraoxosulphate (VI) acid: Industrial Preparation (Contact Process).
HYDROGEN SULPHIDE, H2S
Hydrogen sulphide is found in volcanic gases, sulphur springs, coal gas and natural gas.
LABORATORY PREPARATION
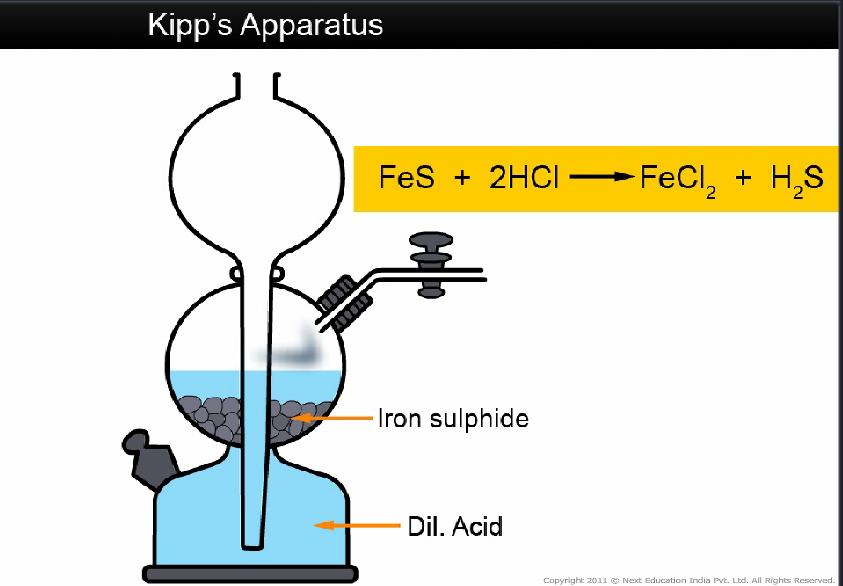
Hydrogen sulphide is prepared in the laboratory by the action of dilute acids on metallic sulphide like Iron (II) sulphide
2HCl(aq) + FeS(s) → FeCl2(aq) + H2S(g)
The apparatus used for a regular supply of hydrogen sulphide in the laboratory is Kipp’s apparatus.
PHYSICAL PROPERTIES
- Hydrogen sulphide is a colourless gas with a smell like that of rotten egg.
- It is very poisonous.
- It is about 1.18 times denser than air.
- It is moderately soluble in water to form a very weak acidic solution.
- It burns with a pale blue flame.
CHEMICAL PROPERTIES
- As an acid, it reacts with alkali to form a normal salt and water
2NaOH(aq) + H2S(g) → Na2S(aq) + 2H2O(l)
- It reacts with excess oxygen to form sulphur (VI) oxide but forms deposit of sulphur with limited oxygen
2H2S(g) + 3O2(g) → 2H2O(l) + 2SO2(g)
2H2S(g) + O2(g) → 2H2(l) + 2S(s)
3. As a reducing agent, it reacts with many oxidizing agents such as acidified KMnO4, acidified K2Cr2O7, chlorine gas, FeCl2, SO2, H2SO4 and HNO3
TEST FOR HYDROGEN SULPHIDE
A piece of filter paper is moistened with lead (II) trioxonitrate (V) solution and dropped it into a gas jar of the unknown gas. If the paper turns black, then the gas is H2S.
EVALUATION
- Name the reagents used in the laboratory preparation of hydrogen sulphide.
- List TWO physical and chemical properties each of hydrogen sulphide.
SULPHUR (IV) OXIDE, SO2
LABORATORY PREPARATION
Sulphur (IV) oxide is prepared in the laboratory by heating sodium or potassium trioxosulphate (IV) with tetraoxosulphate (IV) acid or hydrochloric acid.
Na2SO3(aq) + 2HCl(aq) → 2NaCl(aq) + H2O(l) + SO2(g)
PHYSICAL PROPERTIES
- Sulphur (IV) oxide is a colourless poisonous gas with a smell like that of burning matches.
- It is very soluble in water.
- It is about 2.5 times denser than air.
CHEMICAL PROPERTIES
- As an acid, it reacts with alkali to form normal salt of trioxosulphate and water only
2NaOH(aq) + SO2(g) → Na2SO3(aq) + H2O(l)
- As a reducing agent, sulphur (IV) oxide reacts with many oxidizing agents such as acidified KMnO4; acidified K2Cr2O7; FeCl3, HNO3, chlorine gas. It decolorizes acidified purple KMnO4 and turns acidified orange K2Cr2O7 to green.
- It reacts as bleaching agent decolourising dye by its bleaching action. The bleaching action is similar to that of chlorine in that there must be water. But, while chlorine bleaches by oxidation, sulphur IV oxide bleaches by reduction.
- Sulphur (IV) oxide reacts as an oxidizing agent in the presence of a strong reducing agent such as hydrogen sulphide.
2H2S(g) + SO2(g) → 2H2O(l) + 3S(s)
C(s) + SO2(g) → CO2(g) + S(s)
TEST FOR SO2
- If an unknown gas bleaches a coloured flower, SO2 can be suspected
- The unknown gas bubbled through a solution of either acidified potassium heptaoxodichromate (VI) or potassium tetraoxomanganate (VII). If the orange colour of the acidified K2Cr2O7 turn green or the purple colour of the acidified KMnO4 turns colourless, then the unknown gas is SO2
USES
- It is used in the manufacture of tetraoxosulphate (VI) acid.
- It is used as a germicide and a fumigant especially for destroying termites.
- It is used as a bleaching agent for straw, silt and wood.
- It is used as a preservative in some liquid e.g orange juice.
- Liquid sulphur (IV) oxide is used as a refrigerant.
EVALUATION
- Give one method of preparing sulphur (IV) oxide in the laboratory.
- State THREE physical and chemical properties of sulphur (IV) oxide.
SULPHUR (VI) OXIDE, SO3
Sulphur (VI) oxide is prepared by reacting sulphur (IV) oxide and oxygen under special conditions which are:
- Presence of platinized asbestos or vanadium (V) oxide as catalyst
- Pressure of 1atm
- Temperature range of 400oC – 450o
2SO2(g) + O2(g) 2SO3(g)
PHYSICAL PROPERTIES OF SO3
- It exists as white needle-like crystals at room temperature.
- It has a low boiling point and vaporizes on gentle heating.
- It dissolves readily in water to give tetraoxosulphate (VI) acid.
TRIOXOSULPHATE IV ACID, H2SO3
Trioxosulphate (IV) acid is a dibasic acid with a molecular formula H2SO3
LABORATORY PREPARATION OF H2SO3
It is prepared by the action of dilute hydrochloric acid on heated sodium trioxosulphate (IV) to produce sulphur (IV) oxide, which is then dissolved in water.
Na2SO3(s) + 2HCl(aq) → 2NaCl(aq) + H2O(l) + S02(g)
H2O(l) + SO2(g) H2SO3(aq)
Sulphur (IV) Oxide is the acid anhydride of trioxosulphate (IV) acid.
PHYSICAL PROPERTIES OF H2SO3
- It turns blue litmus paper red.
- It is a colourless liquid.
- It mixes readily with water.
- It has an irritating and choking smell.
CHEMICAL PROPERTIES OF H2SO3
- It reacts with alkalis to form salt and water.
2NaOH(aq) + H2SO3(aq) → Na2SO3(aq) + 2H2O(l)
- It is oxidized in air to tetraoxosulphate (VI) acid
2H2SO3(aq) + O2(g) → 2H2SO4(aq)
- It reduces oxidizing agent such as potassium tetraoxomanganate (VII) and potassium heptaoxodichromate (VI).
- It bleaches dyes in the presence of water.
TEST FOR SO32-
Barium chloride solution is added to the solution of the unknown substance. A white precipitate soluble in dilute hydrochloric acid confirms the presence of a trioxosulphate (IV) ion.
USES OF H2SO3
- It is used for bleaching straw and other fabrics.
- It is used as a germicide.
EVALUATION
- Write an equation of reaction for the laboratory preparation of trioxosulphate IV
- State two physical and two chemical properties of trioxosulphate IV
TETRAOXOSULPHATE VI ACID, H2SO4
Tetraoxosulphate VI acid is one of the most important chemical compounds known. It is used in almost every manufacturing process; hence it is mostly prepared industrially.
INDUSTRIAL PREPARATION OF H2SO4
Industrially, tetraoxosulphate VI acid is manufactured by Contact process. The following steps are involved in the Contact process.
- Burning sulphur in dry air to obtain sulphur (IV) oxide, SO2
S(s) + O2(g) → SO2(g)
- The sulphur (IV) oxide produced is mixed with excess air and passed through an electric chamber to remove impurities and dust which may poison the catalyst. The gaseous mixture is then passed through concentrated H2SO4 to dry it before passing it into the reaction chamber.
- The dried gaseous mixture is delivered to the contact tower (reaction chamber) where the sulphur (IV) oxide and oxygen combine in the presence of pellets of catalyst, vanadium (V) oxide, V2O5 to yield sulphur (VI) oxide. The reaction takes place at atmospheric pressure and temperature of 450-500o
2SO2(g) + O2(g) 2SO3(g) + heat
- The sulphur (VI) oxide is cooled and then dissolved in concentrated H2SO4 to produce a thick liquid called Oleum.
H2SO4(aq) + SO3(g) → H2S2O7(aq)
- The Oleum is then diluted with water appropriately to produce 98% tetraoxosulphate (VI) acid.
H2O(l) + H2S2O7(aq) → 2H2SO4(aq)
NOTE: Sulphur (VI) oxide is not dissolved directly in water to produce the acid because of the large amount of heat that is evolved in the process. The heat is capable of boiling the acid formed to produce a mist of droplets which can spread throughout the factory and cause acid burns.
PHYSICAL PROPERTIES
- It is a colourless, viscous liquid with a density of 1.84gcm-3
- It is corrosive and causes burns when in contact with the skin.
- It is highly soluble in water evolving a large amount of heat.
CHEMICAL PROEPERTIES
- As an acid, it reacts with metal which are above hydrogen in the electrochemical series to liberate hydrogen gas
Zn(s) + H2SO4(aq) → ZnSO4(aq) + H2(g)
- It reacts with bases to form salts and water
MgO(s) + H2SO4(aq) → MgSO4(aq) + H2O(l)
- It reacts with alkali to form normal and acidic salt
H2SO4(aq) + NaOH(aq) → NaHSO4(aq) + H2O(l)
H2SO4(aq) + NaOH(aq) → Na2 SO4(aq) + 2H2O(l)
- As an acid, it reacts with trioxocarbonate (IV) to liberate carbon (IV) oxide
H2SO4(aq) + CuCO3(aq) → CuSO4(aq) + H2O(l) + CO2(g)
- As an oxidizing agent, concentrated H2SO4 oxidize metals, non –metals and hydrogen sulphide in the following ways.
Zn(s) + 2H2SO4(aq) → ZnSO4(aq) + 2H2O(l) + SO2(g)
C(s) + 2H2SO4(aq) → 2H2O(l) + CO2(g) + 2SO2(g)
H2SO4(aq) + H2S(g) → S(s) + H2O(l) + SO2(g)
- Concentrated tetraoxosulphate (VI) acid is a dehydrating agent, removing water from compounds like sugar, ethanol, methanoic acid and ethanedioic acid
C12H22O11(s) → 12C(s) + 11H2O(l)
sugar charcoal
- Concentrated tetraoxosulphate (VI) displaces volatile acids from their salts
KCl(s) + H2SO4(aq) → KHSO4(aq) + HCl(g)
TEST FOR SO42-
Barium chloride solution is added to the solution of the unknown substance. A white precipitate insoluble in excess dilute hydrochloric acid confirms the presence of a tetraoxosulphate (VI) ion.
USES OF H2SO4
- It is used in the production of fertilizers e.g. ammonium tetraoxosulphate (VI).
- It is used in the purification of crude oil.
- It is used as an electrolyte in a lead-acid accumulator.
- It is used as a drying agent for many gases except NH3 and H2S gas.
- It is used to clean metals before electroplating.
USES OF TETRAOXOSULPHATE (VI) SALTS
- Ammonium tetraoxosulphate (VI) used as fertilizers
- Sodium tetraoxosulphate (VI) is used in paper manufacture and as a purgative
- Calcium tetraoxosulphate (VI) is mined as gypsum and when heated forms plaster of Paris used to set broken bones.
- Aluminium tetraoxosulphate (VI) is used to coagulate precipitate in the purification of water
- Iron II tetraoxosulphate (VI) is used to treat anaemia.
We have come to the end of this class. We do hope you enjoyed the class?
Should you have any further question, feel free to ask in the comment section below and trust us to respond as soon as possible.
We have come to the end of this term. It’s been a remarkable journey and we are glad that you have made it this far. For making it this far, we commend you for being resilient, you have taken charge of your education and future.
The Journey still continues though, we are moving on to SS3. we hope to meet you there.
