Back to: CHEMISTRY SS2
Welcome to SS2 Third Term!
We are eager to have you join us in class!!
In today’s Chemistry class, We will be discussing Water. We hope you enjoy the class!
CONTENT
- Sources, Types, Uses and Structure of Water.
- Laboratory Preparation of Water.
- Test for Water
- Causes/ Removal of Hardness of Water.
- Purification of Water for Municipal Supply.

WATER
Water is regarded as the universal solvent. It is a good solvent for many substances.
SOURCES OF WATER
The following are the sources of water:
- Natural water: Rainwater, Well water, Springwater and Seawater
- Treated water: Distilled water, Pipe – borne water and chlorinated water.
TYPES OF WATER
Water is of two types namely: soft water and hard water.
Soft water forms lather with soap easily while hard water does not form lather readily with soap since it contains some dissolved salt in it.
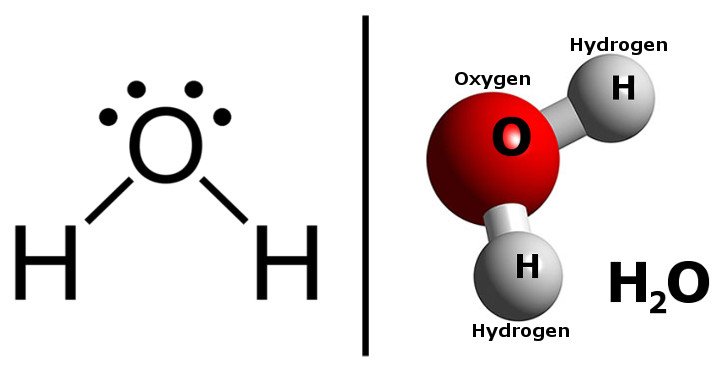
STRUCTURE OF WATER
In a molecule of water, H2O, the central atom is Oxygen. Oxygen has the following electronic configuration: 1s2 2s2 2p4.
The valence shell of oxygen has two lone pairs of electrons (2s22p2) and two unpaired electrons (2py12pz1). Each unpaired electron forms a covalent bond with an electron from a hydrogen atom. The water molecule has two lone pairs and two bond pairs of electrons in the valence shell of its central atom, thereby satisfying the octet rule for stability.
Ideally, the four-electron pairs should be directed towards corners of a tetrahedron. However, when lone pairs of electrons are located near another lone pair, the repulsion between them is so great that they squeeze the other two bond pairs of electrons closer together. As a result, the bond angle in water is compressed to approximately 105o, such that the structure of the water molecule is V-shaped or angular shape.
LABORATORY PREPARATION OF WATER
To prepare water in the laboratory, dry hydrogen gas is ignited in air. It burns with a faint blue flame to give steam, which will condense on contact with any cold surface to form water.
PHYSICAL PROPERTIES OF WATER
- Water boils at 100oC and freezes at 0oC
- It has a maximum density of 1gcm-3 at 4oC
- It is neutral to litmus.
CHEMICAL PROPERTIES
- Water reacts with electropositive metals to form alkali and liberate hydrogen gas. E.g
Na(s) + H2O(aq) NaOH(aq) + H2(g)
Mg & Zn react with steam
Cu, Au, Ag, Hg do not react with water to form an alkaline solution
- Non-metal like chlorine reacts with water to form acid solution.
H20(aq) + Cl2(g) HCl(aq) + HOCl(aq)
TEST FOR WATER
When a few drops of water are added to
- White anhydrous copper (II) tetraoxosulphate (VI), it turns blue.
- Blue cobalt (II) chloride, it turns pink.
NOTE: These two tests are not specific for water. They only indicate the presence of water. Any aqueous solution or substance containing water will give a positive test for water.
SELF EVALUATION
- Describe the structure of water.
- How will you identify a give solution to be water?
HARDNESS OF WATER
Hard water is the water that does not form lather readily with soap.
Water acquired hardness when insoluble salts of CaSO4, MgSO4 and Ca(HCO3)2 dissolves in it from the soil which it flows through.
TYPES OF HARDNESS OF WATER
- Temporary hard water
- Permanent hard water
TEMPORARY HARDNESS: This is caused by the presences of Ca2+ and Mg2+ in the form of hydrogen trioxocarbonate IV i.e. Ca(HCO3)2
REMOVAL OF TEMPORARY HARDNESS
- Physical method: By boiling
Ca(HCO3)2(aq )heat CaCO3(s) + H2O(l) + CO2(g)
- Chemical method: By using of slaked lime (calcium hydroxide solution)
Ca(HCO3)2(aq) + Ca(OH)2(aq) 2CaCO3(s)+ 2H2O(l)
EFFECTS OF TEMPORARY HARDNESS: It causes
- Furring of kettles and boilers.
- Stalagmite and stalactites in caves.
PERMANENT HARDNESS
Permanent hardness in water is caused by the presence of Calcium and Magnesium ions in the form of soluble tetraoxosulphate (VI) and chlorides (i.e. CaSO4, MgSO4, MgCl2, CaCl2)
Removal of permanent hardness: By chemical method only
- Addition of washing soda
Na2CO3(aq) + CaSO4(aq) CaCO3(s) + Na2SO4(aq)
- Addition of caustic soda
2NaOH(aq) + CaSO4(aq) Ca(OH)2(s) + Na2SO4(aq)
- Ion exchange resin
CaSO4(aq) + Sodium zeolite Calcium zeolite + NaSO4(aq)
(insoluble)
ADVANTAGES OF HARD WATER
- It has better taste than soft water.
- Calcium salts in it help to build strong teeth and bones.
- It provides CaCO3, that crab and snail use to build their shells.
- It does not dissolve lead, hence it can be supplied in lead pipes.
DISADVANTAGES OF HARD WATER
- It causes furring of kettles and boilers.
- It wastes soap.
- It cannot be used in dyeing and tanning.
SELF EVALUATION
- Mention TWO compounds that can cause permanent hardness of water.
- Write two equations to show the removal of permanent hardness of water.
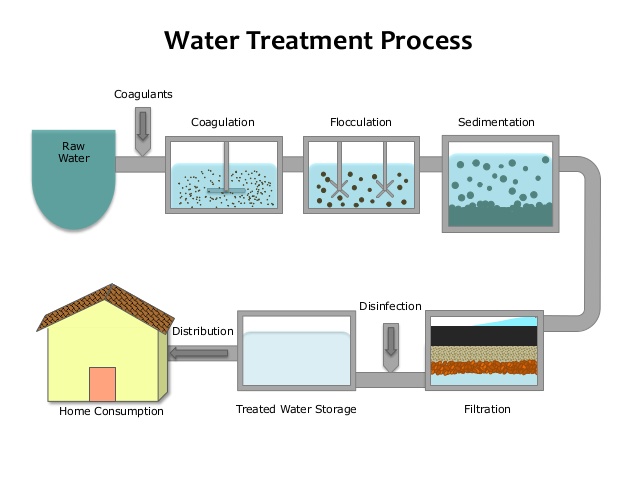
TREATMENT OF WATER FOR MUNICIPAL SUPPLY
The following are the processes of treating river water for town supply
- Coagulation: Chemicals like potash alum, KAl(SO4)2, or sodium aluminate III, NaAlO2 is added to water in a large settling tank.
- Sedimentation: The coagulated solid particles or flocs are allowed to settle in the settling tank to form sediments at the bottom of the tank.
- Filtration: The water above the sediment still contains some suspended particles. The water is passed through a filter bed to remove the remaining fine dirt particles.
- Chlorination (Disinfection): Chemicals like chlorine is then added to the water to kill germs. Iodine and fluorine are also added as food supplements to prevent goitre and tooth decay respectively. The treated water is then stored in a reservoir and distributed to the town.
GENERAL SELF EVALUATION/REVISION
- Mention two compounds that causes permanent hardness in water
- State two ways of removing permanent hardness in water
- List two advantages of hard water
- State Faraday’s second law of electrolysis
- Using electron dot-cross representation, show the formation of carbon (IV) oxide and name the type of bond formed
READING ASSIGNMENT
New School Chemistry for Senior Secondary School by O.Y.Ababio (6th edition) pages 296-302
WEEKEND ASSIGNMENT
SECTION A: Write the correct option ONLY
- Treated town water undergoes the following steps except A. coagulation precipitation C. sedimentation D. chlorination
- Water is temporarily hard because it contains A. CaSO4 MgSO4C. chlorine D.Ca(HCO3)2
- Temporary hardness of water is removed by the use of one of the following A. boiling use of Ca(OH)2 C. use of Na2CO3 D. use of alum
- A substance that turns white anhydrous CuSO4 blue is A. water liquid ammonia C. hydrochloric acid D. molten sulphur
- Distilled water is different from deionized water because A. distilled water is a product of condensed steam while deionized water is filtered laboratory water distilled water is always pure and sold in packs while deionized is not packaged for consumption C. distilled water is condensed steam but deionized water is produced using ion-exchange resins which absorbs undesired ions. D. distilled water is man-made while deionized water is both natural and artificial
SECTION B
- State the steps involved in the treatment of river water for town supply.
- Write two equations to show the removal of permanent hardness of water.
We have come to the end of this class. We do hope you enjoyed the class?
Should you have any further question, feel free to ask in the comment section below and trust us to respond as soon as possible.
In our next class, we will be talking about Solubility and Solutions. We are very much eager to meet you there.
