Back to: CHEMISTRY SS2
Welcome to Class !!
We are eager to have you join us !!
In today’s Chemistry class, We will be learning about Mass / Volume Relationship. We hope you enjoy the class!
CONTENT
- Mole and Molar Quantities
- Relative Atomic Mass and Relative Molecular Mass.
- Calculations involving Mass and Volume.
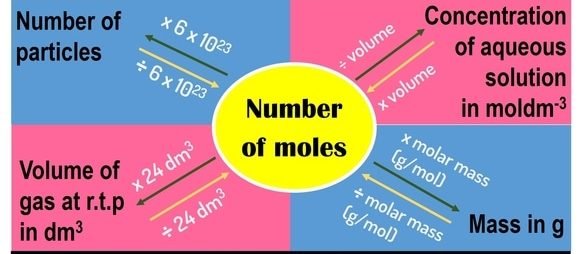
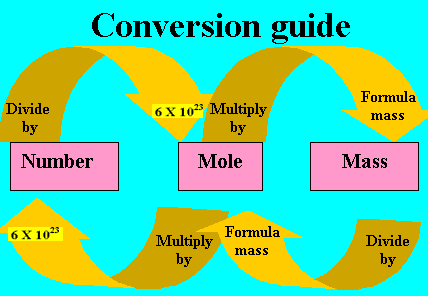
MOLE AND MOLAR QUANTITIES
THE MOLE
A mole is a number of particles of a substance which may be atoms, ions, molecules or electrons. This number of particles is approximately 6.02 x 1023 in magnitude and is known as Avogadro’s number of particles.
The mole is defined as the amount of a substance which contains as many elementary units as there are atoms in 12g of Carbon-12.
RELATIVE ATOMIC MASS
The relative atomic mass of an element is the number of time the average mass of one atom of that element is heavier than one-twelfth the mass of one atom of Carbon-12. It indicates the mass of an atom of an element. For e.g, the relative atomic mass of hydrogen, oxygen, carbon, sodium and calcium are 1, 16, 12, 23, and 40 respectively.
The atomic mass of an element contains the same number of atoms which is 6.02 x 1023atoms; 1 mole of hydrogen having an atomic mass of 2.0g contains 6.02 x 1023 atoms.
RELATIVE MOLECULAR MASS
The relative molecular mass of an element or compound is the number of times the average mass of one molecule of it is heavier than one-twelfth the mass of one atom of Carbon-12
It is the sum of the relative atomic masses of all atoms in one molecule of that substance. It is also called the formula mass. The formula mass refers not only to the relative mass of a molecule but also that of an ion or radical.
CALCULATION
Calculate the relative molecular mass of:
- Magnesium chloride
- Sodium hydroxide
- Calcium trioxocarbonate
[Mg=24, Cl=35.5, Na=23, O=16, H=1, Ca=40,C=12]
Solution:
- MgCl 2 = 24 + 35.5×2 = 24 + 71 = 95gmol-1
- NaOH = 23 + 16 + 1 = 40gmol-1
- CaCO3 = 40 + 12 +16×3 = 100gmol-1
EVALUATION
- What is the relative molecular mass of a compound?
- Calculate the relative molecular mass of (a) NaNO3 (b) CuSO4.5H2O
MOLAR VOLUME OF GASES
The volume occupied by 1 mole of a gas at standard conditions of temperature and pressure (s.t.p) is 22.4 dm3. Thus 1 mole of oxygen gas of molar mass 32.0gmol-1 occupies a volume of 22.4dm3 at s.t.p and 1 mole of helium gas of molar mass 4.0gmol-1 occupies a volume of 22.4 dm3 at s.t.p.
Note: When the conditions of temperature and pressure are altered, the molar volume will also change. Also, standard temperature = 273K and standard pressure = 760mmHg.
RELATIONSHIP BETWEEN QUANTITIES
Note: Amount = Number of moles
CALCULATIONS
- What is the mass of 2.7 mole of Aluminium (Al=27)?
Solution:
Reacting mass = Amount x Molar mass
= 2.7mole x 27 gmol-1 = 72.9g.
- What is the number of oxygen atoms in 32g of the gas? (O=16, NA = 6.02 x 1023)
Solution:
EVALUATION
- Define the molar volume of a gas
- How many molecules are contained in 1.12dm3 of hydrogen gas at s.t.p.?
STOICHIOMETRY OF REACTION
The calculation of the amounts (generally measured in moles or grams) of reactants and products involved in a chemical reaction is known as the stoichiometry of the reaction. In other words, the mole ratio in which reactants combine and products are formed gives the stoichiometry of the reactions.
From the stoichiometry of a given balanced chemical equation, the mass or volume of the reactant needed for the reaction or products formed can be calculated.
CALCULATION OF MASSES OF REACTANTS AND PRODUCTS
- Calculate the mass of solid product obtained when 16.8g of NaHCO3was heated strongly until there was no further change.
Solution:
The equation for the reaction is:
2NaHCO3(s) → Na2CO3(s) + H2O(g) CO2(g)
Molar mass of NaHCO3 = 23 + 12 + 16×3 = 84gmol-1
Molar mass of Na2CO3 = 23×2 +12+16×3 = 106gmol-1
From the equation:
2 moles NaHCO3 produces 1 mole Na2CO3
2x84g NaHCO3 produces 106g Na2CO3
16.8g NaHCO3 will produce Xg Na2CO3
Mass of solid product obtained = 10.6g
- Calculate the number of moles of CaCl2 that can be obtained from 25g of limestone [CaCO3] in the presence of excess acid.
Solution:
The equation for the reaction is:
CaCO3(s) + 2HCl → CaCl2(s) + H20(l) + CO2(g)
Molar mass of CaCO3 = 40 + 12 + 16×3 = 100gmol-1
From the equation of reaction,
1 mole CaCO3 yields 1 mole CaCl2
Therefore, 0.25 mole CaCO3 yielded 0.25 mole CaCl2.
EVALUATION
- What does the term ‘Stoichiometry of reaction’ mean?
Ethane [C2H6] burns completely in oxygen. What amount in moles of CO2will be produced when 6.0g of ethane is completely burnt in oxygen?
CALCULATION OF VOLUME OF REACTING GASES
- In an experiment, 10cm3 of ethene [C2H4] was burnt in 50cm3 of oxygen.
-
- A. Which gas was supplied in excess? Calculate the volume of the excess gas remaining at the end of the reaction.
- B. Calculate the volume of CO2 gas produced
-
Solution:
The equation for the reaction is:
C2H4(g) + 3O2(g) → 2CO2(g) + 2H2O(g)
A. From the equation,
1 mole of ethene reacts with 3mole of oxygen
1 volume of ethene reacts with 3 volumes of oxygen
10cm3 of ethene will react with 30cm3 of oxygen
Since 50cm3 of oxygen was supplied, oxygen was in excess
Hence volume of the excess gas = initial volume – volume used up = 50-30 = 20cm3
B. 1 volume of ethene produces 2 volumes of CO2
10 cm3 of ethene will produce 20cm3 of CO2
Therefore, 20cm3 of CO2 was produced
- 20cm3 of CO was mixed and sparked with 200cm3 of air containing 21% of O2. If all the volumes are measured at s.t.p, calculate the total volume of the resulting gases.
Solution:
In 200cm3 of air,
Volume of O2 = 21 x 200cm3 = 42cm3
100
Volume of N2 and rare gases = 200-42 = 158cm3
The equation for the reaction is:
2CO(g) + O2(g) → 2CO2(g)
Volume ratio 2 : 1 : 2
Before sparking 20cm3 42cm3
Reacting volume 20cm3 10cm3
After sparking 32cm3 20cm3
Volume of resulting gases = 32 + 20 + 158 = 210cm3
GENERAL EVALUATION/REVISION
- Find the volume of oxygen produced by 1 mole of KClO3 at s.t.p. in the following reaction: 2KClO3(s) → 2KCl(s) + 302(g)
- Define the term ‘Relative atomic mass’
- Balance the following redox equations I– + MnO4– IO3– + MnO2 in basic medium
- Write the symbols of the following elements: mercury, silver, gold, lead, tin, antimony.
- Define valency.
READING ASSIGNMENT
New School Chemistry for Senior Secondary School by O. Y. Ababio, Pg 156-164
WEEKEND ASSIGNMENT
SECTION A: Write the correct option ONLY
- Amount of a substance is expressed in a. mole b. grams c. kilograms d. mass
- Determine the mass of CO2 produced by burning 104g of ethyne [C2H2] a. 256g b.352g c. 416g d. 512g
- The mole ratio in which reactants combine and products are formed is known as a. rate of reaction b. stoichiometry of reaction C. equation of reaction d. chemical reaction
- The unit for relative molecular mass is A. mole B. gmol-1 C. grams D. mass
- What mass of Pb(NO3)2 would be required to 9g of PbCl2 on the addition of excess NaCl solution? [Pb=207, Na=23, O=16, N=14] A. 10.7g B. 1.2g C. 6.4g D. 5.2g
SECTION B
- Calculate the number of molecules of CO2 produced when 10g of CaCO3 is treated with 100cm3 of 0.20moldm-3HCl
- Calculate the volume of nitrogen that will be produced at s.t.p. from the decomposition of 9.60g ammonium dioxonitrate (III), NH4NO2.
We have come to the end of this class. We do hope you enjoyed the class?
Should you have any further question, feel free to ask in the comment section below and trust us to respond as soon as possible.
In our next class, we will be learning about Acid-Base Reactions. We are very much eager to meet you there.
