Back to: CHEMISTRY SS3
Welcome to class!
In today’s class, we will be talking about Alkanols. Enjoy the class!
Alkanols
Sources of alkanols
- From destructive distillation wood.
- From starchy food and sugar
General molecular formula
Alkanol is a homologous series with the general molecular formula of CnH2n+1OH or ROH or (CnH2n+2O).
Nomenclature
The names of alkanols are obtained by substituting “e” in alkanes with “ol” in alkanol e.g. methanol (CH3OH), ethanol (CH3CH2OH).
Classification
The alkanols are classified based on the number of alkyl groups directly linked to the carbon atom holding the hydroxyl group.
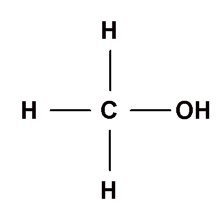
- Primary alkanols (10): It has only one alkyl group attached to the carbon atom that carries the hydroxyl group e.g.

- Secondary alkanols (20): They have 2 alkyl groups directly linked with the carbon atom holding the hydroxyl group e.g.
- Tertiary alkanols (30 ): the alkanols here have 3 alkyl groups attached to the carbon atom holding the hydroxyl group
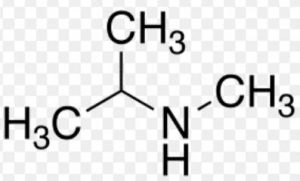
The R1, R2 are alkyl groups which may be the same or not.
N.B: Examine the structure of the alkanol, identify the carbon atom bonded to the –OH, count the number of alkyl groups that are directly attached to the carbon atom. Then you can say whether it is 10, 20 or 30.
Evaluation
- Name the functional group in the alkanol.
- Give an example each by writing the structure and names of the classes of alkanols.
Types of alkanol
The type of alkanols is determined by the number of the hydroxyl group –OH, present in the molecule.
- Monohydric alkanols: This type has only one –OH, present in its molecule e.g. C2H5OH, C3H7OH e.t.c.
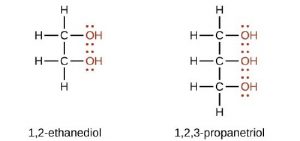
- Dihydric alkanols: This type has two of –OH group per molecule e.g.
- Polyhydric alkanols: This type has three or more. OH groups per molecule e.g. glycerol.
Preparation e.g. ethanol
- Laboratory preparation
- Hydrolyzing ethyl esters with hot alkali
- Reducing ethanol with nascent hydrogen
- Commercial preparation
- From ethene
Ethene is obtained by the cracking of petroleum. It is then absorbed in 95% H2SO4 at 800C and
30 atoms to form C2H5HSO4
C2H4 + H2SO4 → C2H3HSO4
The C2H3HSO4 is bedded in water to produce ethanol;
C2H3HSO4 + H2O → C2H3OH + H2SO4.
The ethanol is distilled off leaving the acid behind which can be used again.
- Preparation by fermentation ethanol is prepared industrially from raw materials containing starch or sugar by the process of fermentation.
- The fermentation is an enzymatic process which involves the decomposition of large organic molecules to a simple molecule by micro-organism.
- The common micro-organism used is YEAST
Evaluation
- Name three (3) types of alkanol and give one example each.
- State two methods by which ethanol can be prepared industrially.
Production of ethanol from starchy materials (e.g. rice, potatoes, maize etc.)
The following steps are involved;
- Crush and pressure cook the starchy materials.
- Extract the starch granules by mixing with water.
- Allow the starch granules to settle and decant
- Treat the starch granules with malt (a partially germinated barley which contains the enzyme, DIASTASE) at 500C for I hour.
- The starch is then converted to MALTOSE; 2(C6H10O5)n + nH2O nC12H22O11
Then add yeast at room temperature for some time (at least one day). Yeast contains two enzymes, namely MALTASE and ZYMASE. Maltase converts maltose to two glucose units, while zymase converts the glucose to ethanol and carbon (iv) oxide.
C12H22O11 + H2O maltase → 2C6H12O6
C6 H12 O6 Zymase → 2C2H3OH + CO2
Ethanol
Evaluation
- Describe fully, the production of ethanol from a named starchy material/food.
- What type of chemical reaction is involved in the fermentation of sugar?
Physical properties
- Ethanol is a colourless volatile liquid.
- It is soluble in water.
- It has a boiling point of 780C.
- It has no action on litmus paper.
Chemical properties
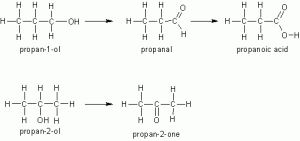
- Combustion: the lower members of alkanols burn with clean flames in plenty of air
2CH3OH + 3O2 → 2CO2 4 +H2O
- Oxidation:
The products of oxidation depend on the structure of the alkanol.
- Primary alkanols are oxidized to alkanol first and then to alkanoic acid in the presence of oxidizing agent e.g KNnO4
CH3CH2OH O CH3CHO O CH3COOH
- Secondary alkanols oxidize to ketone e.g.
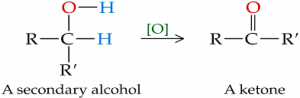
Tertiary alkanols are not oxidized because there is no carbon-hydrogen bond to be broken for the oxidation to take place.
N.B: Note the colour change of oxidizing agent if acidified.
- Purple KMnO4 changes to colourless.
- Orange K2Cr2O7 turns green.
- Esterification:
This is the reversible reaction between alkanol and alkanoic and to produce an ester. The reaction is catalyzed by conc. H2SO4 e.g. CH3CH2OH + CH2COOCH2CH3 + H2O
- Dehydration:
Alkanols are dehydrated to alkenes in the presence of conc H2SO4
e.g. CH3CH2OH + conc H2SO4 → CH3CH2HSO4 + H2O
CH3CH2HSO 4170 C → C2H4 + H2SO4
- Reaction with sodium and potassium:
sodium and potassium react vigorously with alkanols to liberate hydrogen gas and form corresponding organic salt of sodium and potassium.
E.g. 2C2H3OH + Na → 2C2H3ONa + H2
N.B: If sodium ethoxide reacts with water, it hydrolyses to ethanol and alkaline solution of NaOH.
- Reactions with the chlorides of phosphorus: Ethanol reacts vigorously with PCl5 in the cold to produce a type of HCl and chloroethane vapour
C2H5OH + PCl5 → C2H3Cl + POCl3 + HCl
-PCl3 gives a similar reaction, but less vigorous
C2H5OH + PCL3 → 3C2H5Cl + H3PO3.
Uses of ethanol
- It is used as an organic solvent
- It is the main constituent of the methylated spirit used to clean wounds and to dissolve paint.
- It is used as petrol addictive for use as fuel in vehicles.
- Used to manufacture other chemicals such as ethanol and ethanoic acid.
- It is used as an ingredient in making alcoholic drinks e.g. beers, wines and spirits.
- It is used as anti-freeze in automobile radiator because of its low freezing point (-1170C).
Test for alkanols
- It librates hydrogen gas on reacting with sodium methal.
- It changes to alkanoic acid (primary alkanol) on exposure to air or reacting with an oxidizing agent.
General valuation
- State five (5) chemical properties of ethanol.
- Identify two (2) physical properties of ethanol
- State two (2) uses of alkanol.
Reading assignment
New School Chemistry By O.Y. Osei Yaw Ababio pages 496-501
Weekend assignment
- The functional groups of the alkanol is _______ A. CnH2n + 1 OH B. carboxylic group C. hydroxyl group D. CnH2n+ 2
- Primary alkanols are oxidized to carboxylic acid, secondary alkanols are oxidized to alkanones while tertiary alkanols are A. oxidized to alkanols B. oxidized to alkanones C. not oxidized D. oxidized to alkene
- The soluble of alkanols in water is due to A. the covalent nature B. hydrogen bonding C. their low melting point D. their low melting point
- When acidified KMnO4 is used as oxidizing agent for alkanol, the colour change observed is A. yellow to red B. purple to colourless C. orange to green D. white to black
- Which of the following enzymes converts glucose to ethanol? A. maltose B. zymase C. diastase D. amylase
Theory
1(a) Write the structural formula of two named secondary alkanols.
(b) Explain the structural difference between primary and tertiary alkanols.
2(a) What is fermentation?
(b) Describe the preparation of ethanol from table sugar.
In our next class, we will be talking about Alkanoic Acids. We hope you enjoyed the class.
Should you have any further question, feel free to ask in the comment section below and trust us to respond as soon as possible.
