Back to: PHYSICS SS3
Welcome to class!
In today’s class, we will be talking about atomic models. Enjoy the class!
Atomic Models
In the 18th and 19th centuries, many scientists attempted to explain the structure of the atom with the help of atomic models. Each of these models had their own merits and demerits and were pivotal to the development of the modern atomic model. The most notable contributions to the field were by the scientists J.J. Thomson, Ernest Rutherford, and Niels Bohr.
Thomson atomic model
The English chemist Sir Joseph John Thomson put forth his model describing the atomic structure in the early 1900s.
He was later awarded the Nobel prize for the discovery of “electrons”. His work is based on an experiment called cathode ray experiment. The construction of working of the experiment is as follows:
Cathode Ray Experiment
It has a tube made of glass which has two openings, one for the vacuum pump and the other for the inlet through which a gas is pumped in.
Cathode Ray Experiment – The discovery of electrons
The role of the vacuum pump is to maintain “partial vacuum” inside the glass chamber. A high voltage power supply is connected using electrodes i.e. cathode and Anode is fitted inside the glass tube.
Observations:
- When a high voltage power supply is switched on, rays were emerging from the cathode towards the anode. This was confirmed by the ‘Fluorescent spots’ on the ZnS screen used. These rays were called “Cathode Rays”.
- When an external electric field is applied, the cathode rays get deflected towards the positive electrode, but in the absence of electric field, they travel in a straight line.
- When rotor Blades are placed in the path of the cathode rays, they seem to rotate. This proves that the cathode rays are made up of particles of a certain mass so that they have some energy.
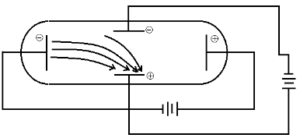
- With all this evidence, Thompson concluded that cathode rays are made of negatively charged particle called “electrons”.
On applying the electric and magnetic field upon the cathode rays (electrons), Thomson found the charge to mass ratio (e/m) of electrons. (e/m) for electron: 17588 × 1011 e/bg.
From this ratio, the charge of the electron was found by Mullikin through oil drop experiment. [Charge of e- = 1.6 × 10-16 C and Mass of e- = 9.1093 × 10-31 kg].
Conclusion
Thomson described the atomic structure as a positively charged sphere into which negatively charged electrons were embedded.
It is commonly referred to as the “plum pudding model” because it can be visualized as a plum pudding dish where the pudding describes to the positively charged atom and the plum pieces describe the electrons.
Thomson’s atomic structure described atoms as electrically neutral, i.e. the positive and the negative charges were of equal magnitude.
Limitations of Thomson’s Atomic Structure: Thomson’s atomic model does not clearly explain the stability of an atom. Also, further discoveries of other subatomic particles, couldn’t be placed inside his atomic model.
Rutherford atomic theory
Rutherford, a student of J. J. Thomson modified the atomic structure with the discovery of another subatomic particle called “Nucleus”. His atomic model is based on the Alpha ray scattering experiment.
Alpha ray scattering experiment
Construction
- A very thin gold foil of 1000 atoms thick is taken.
- Alpha rays (doubly charged Helium He2+) were made to bombard the gold foil.
- ZnS screen is placed behind the gold foil.
Observation
- Most of the rays just went through the gold foil making scintillations (bright spots) in the ZnS screen.
- A few rays got reflected after hitting the gold foil.
- One in 1000 rays got reflected by an angle of 180° (retraced path) after hitting the gold foil.
Conclusion
- Since most rays passed through, Rutherford concluded that most of the space inside the atom is empty.
- Few rays got reflected because of the repulsion of its positive with some other positive charge inside the atom.
- 1/1000th of rays got strongly deflected because of a very strong positive charge in the centre of the atom he called this strong positive charge as “nucleus”.
- He said most of the charge and mass of the atom resides in the Nucleus
Rutherford’s structure of the atom
Rutherford proposed his own atomic structure which is as follows.
- The nucleus is at the centre of an atom, where most of the charge and mass is concentrated.
- Atomic structure is spherical.
- Electrons revolve around the nucleus in a circular orbit, similar to the way planets orbit the sun.
Limitations of Rutherford’s atomic model
- If electrons have to revolve around the nucleus, they will spend energy and that too against the strong force of attraction from the nucleus, a lot of energy will be spent by the electrons and eventually, they will lose all their energy and will fall into the nucleus so the stability of atom is not explained.
- If electrons continuously revolve around the ‘nucleus, the type of spectrum expected is a continuous spectrum. But in reality, what we see is a line spectrum.
Subatomic particles
Protons
- Protons are positively charged subatomic particles. The charge of a proton is 1e, which corresponds to approximately 1.602 × 10-19
- The mass of a proton is approximately 1.672 × 10-24
- Protons are over 1800 times heavier than electrons.
- The total number of protons in the atoms of an element is always equal to the atomic number of the element.
Neutrons
- The mass of a neutron is almost the same as that of a proton i.e. 1.674×10-24
- Neutrons are electrically neutral particles and carry no charge.
- Different isotopes of an element have the same number of protons but vary in the number of neutrons present in their respective nuclei.
Electrons
- The charge of an electron is -1e, which approximates to -1.602 × 10-19
- The mass of an electron is approximately 9.1 × 10-31.
- Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom.
Atomic structure of isotopes
Nucleons are the components of the nucleus of an atom. A nucleon can either be a proton or a neutron. Each element has a unique number of protons in it, which is described by its unique atomic number. However, several atomic structures of an element can exist, which differ in the total number of nucleons.
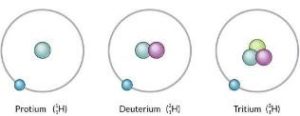
These variants of elements having a different nucleon number (also known as the mass number) are called isotopes of the element. Therefore, the isotopes of an element have the same number of protons but differ in the number of neutrons.
For example, there exist three known naturally occurring isotopes of hydrogen, namely, protium, deuterium, and tritium. The atomic structures of these hydrogen isotopes are illustrated below.
Bohr’s atomic theory
Neils Bohr put forth his model of the atom in the year 1915. This is the most widely used atomic model to describe the atomic structure of an element which is based on Planck’s theory of quantization.
Postulates
- The electrons inside atoms are placed in discrete orbits called “stationery orbits”.
- The energy levels of these shells can be represented via quantum numbers.
- Electrons can jump to higher levels by absorbing energy and move to lower energy levels by losing or emitting its energy.
- As longs as, an electron stays in its own stationery, there will be no absorption or emission of energy.
- Electrons revolve around the nucleus in these stationary orbits only.
- The energy of the stationary orbits is quantized.
Limitations of Bohr’s atomic theory
- Bohr’s atomic structure works only for single-electron species such as H, He+, Li2+, Be3+,
- When the emission spectrum of hydrogen was observed under a more accurate spectrometer, each line spectrum was seen to be a combination of no of smaller discrete lines.
- Both Stark and Zeeman effects couldn’t be explain using Bohr’s theory.
General evaluation
- State the limitations of J.J Thompson Atomic model
Assignment
- Briefly discussed the Postulate and limitations of Neil Bohr’
In our next class, we will be talking about Radioactivity. We hope you enjoyed the class.
Should you have any further question, feel free to ask in the comment section below and trust us to respond as soon as possible.

