Back to: PHYSICS SS3
Welcome to class!
In today’s class, we will be talking about the transformation of elements. Enjoy the class!
Transformation of Elements

Nuclear fission
Nuclear fission is a process in which a nucleus splits into two or more lighter nuclei. This process generates large amounts of energy together with neutron emission. Nearly 80% of the energy produced appears as the kinetic energy of the fission fragments. Thus a chain reaction is set in motion. A chain reaction is a multiplying and self-maintaining reaction. When the size of Uranium exceeds a certain critical mass, there is a rapid production of neutrons accompanied by a release of a tremendous amount of energy in a nuclear explosion. This is the principle of the atomic and nuclear fission bombs. Fission is also the process used in the present-day nuclear power plant.
For example, Uranium-235 undergoes nuclear fission when bombarded with slow neutrons releasing 2-3 neutrons per Uranium molecule and every neutron released brings about the fission of another Uranium-235nuclei.
Another substance which undergoes the same process is Plutonium-239.
Substances which undergo fission directly with slow neutrons are known as fissile substances or isotopes.
Applications of nuclear fission
- They are used in the manufacture of atomic bombs where a tremendous amount of energy is released within a very short time leading to an explosion.
- When this release of energy is controlled such that it can be released at a steady rate then it is converted into electrical energy hence the principle in nuclear reactors.
Nuclear fusion
Nuclear fusion is the thermal combining of light elements to form relatively heavier nuclei. The process requires very high temperatures for the reacting nuclei to combine upon collision.
These temperatures are provided by ordinary fission bombs.
These reactions are sometimes known as thermonuclear reactions.
A fusion reaction releases energy at the rate of 3-23 MeV per fusion event i.e. two deuterium (heavy hydrogen) nuclei to form helium.
![]()
This 3.3 MeV (energy) produced is equal to 5.28 × 10-13 J.
Application of nuclear fusion
- Used in the production of the hydrogen bomb. Possible reactions for a hydrogen bomb include;
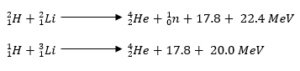
Hazards of radioactivity and their precautions
- Due to the ionizing radiation emitted by radiation materials, they affect living cells leading to serious illnesses. Symptoms of radiation exposures are immature births, deformations, retardedness, etc.
- Their exposure to the environment through leaks may lead to environmental pollution leading to poor crop growth and destruction of marine life.
Applications of radioactivity
- Carbon dating: through the identification of carbon-14 and carbon-12 absorbed by dead plants and animals. Scientists can be able to estimate the age of a dead organism. Since carbon is a radioactive element with a half-life of 5,600 years archaeologists can be able to estimate the ages of early life through carbon dating.
- Medicine: radiation is used in the treatment of cancer, by using a radioactive cobalt-60 to kill the malignant tissue. Radiations are used in taking x-ray photographs using cobalt-60. Radiations are used to sterilize surgical instruments in hospitals. Radioactive elements can also be used as tracers in medicine where they determine the efficiency of organisms such as kidneys and thyroid glands.
- Biology and agriculture: radioactive sources are used to generate different species of plants with new characteristics that can withstand diseases and drought. Insects are sterilized through radiation to prevent the spread of pests and diseases. Potatoes exposed to radiation can be stored for a long time without perishing.
- Industry: the thickness of metal sheets is measured accurately using radiation from radioactive sources. Recently the manufacture of industrial diamonds is undertaken through transmutation.
- Energy source: in N. America, Europe and Russia nuclear reactors are used to generate electricity.
The amount of fuel used is quite small hence an economical way of generating electrical energy as compared to H.E.P generation.
Nuclear energy
This is the energy that is released during nuclear activities.
In a nuclear reaction, the masses of the products are usually different from the masses of the 4 reactants.
Nuclear energy equation:

Electron Volt (EV)
An electron volt is energy possessed by an electron that is moving under the influence of a potential difference of one volt. It is the unit of energy in nuclear energy.
Value of electron volt (ev):
1ev = 1.602 x 10⁻¹⁹ Joule
1Mev = 1.602 x 10⁻¹⁹ Joule x 10⁻6 = 1.602 x 10⁻¹³ Joule
Atomic mass unit (à.m.u.)
The unit of energy is also expressed in a.m.u.
Value of a.m.u:
1a.m.u = 931Mev which will be 931 x 1.602 x 10⁻¹³ Joule
= 1491.462 Joules
Worked examples
1. Calculate the energy of 2.25ev.
Solution
1ev = 1.602 x 10⁻¹⁹
Therefore,
2.25ev will equal 2.25 x 1.602 x10⁻¹⁹ = 3.6045 x 10⁻¹⁹ Joules
2. What is the value of 3.43 Joules in a.m.u?
Solution
1 a.m.u = 931Mev = 931 x 1.602 x 10⁻¹³ = 3.6045 = 10⁻¹⁹ Joules
Therefore,
6874.23 x 10⁻¹⁹ Juole = 0.952 a.m.u
Sources of radiations
- Radioactive fallout from nuclear plants and other sources
- Disposal of nuclear waste.
The biological effect of radiations
- Short term recoverable effect: Diarrhoea, sore throat, loss of hair, nausea, etc.
- Long term irrecoverable effect: Leukemia and cancer, Genetic effects
Safety rules for handling radioactive materials
- People working with radioactive materials should put on special lead-lined aprons and lead gloves.
- They should handle radioactive materials with long lead tongs.
- They should not go beyond the safety limit for each type of radiation.
- The radioactive materials must be kept in thick lead containers with a very narrow opening to stop radiations coming out from other directions.
Safety rules in the establishment of nuclear power plants
- The nuclear reactor of the power plant must be shielded with lead and steel walls.
- The nuclear reactor must be housed in an airtight building of a strong concrete structure.
- There must be a back-up for the cooling system for the reactor core.
- There should be the safe disposal of nuclear waste.
General evaluation
- Define a chain reaction.
Assignment
- Explain the terms of nuclear fission and nuclear fusion.
In our next class, we will be talking about Quantum. We hope you enjoyed the class.
Should you have any further question, feel free to ask in the comment section below and trust us to respond as soon as possible.
