Back to: PHYSICS SS3
Welcome to class!
In today’s class, we will be talking about the conduction of electricity through gases. Enjoy the class!
Conduction of Electricity Through Gases

When voltage is applied across an anode and a cathode that are enclosed in a vacuum which contains gas and the gas is subjected to a reduced or low pressure, the gas conduct electricity.
Conditions under which gases conduct electricity
Gases conduct electricity under low pressure and high voltage.
Explanation of operations
When the pressure inside the tube is reduced to very low pressure ( say -0.01 mm Hg) and a high voltage ( say 1000 volt) is connected across the cathode and the anode of the tube, the gas in tube breaks into ions ( I.e ionized ). The positive ions move toward the cathode while the negative ions and free electrons move toward the anode. At the cathode, the positive ions knock off electrons from the cathode. These electrons produced at the cathode are called cathode rays.
Cathode rays
Cathode rays are a stream of electrons.
Production of cathode rays
Cathode rays are produced when the pressure inside a cathode ray tube is reduced to very low pressure ( say -0.01 mm Hg) and a high voltage ( say 1000 volt) is connected across the cathode and the anode of the tube, the gas in the tube breaks into ions ( I.e ionized ). The positive ions move toward the cathode while the negative ions and free electrons move toward the anode. At the cathode, the positive ions knock off electrons from the cathode. These electrons produced at the cathode are called cathode rays.
Properties of cathode rays
- Cathode rays are a stream of moving particles.
- Cathode rays are negatively charged particles.
- Cathode rays are attracted toward the anode in an electric field.
- Cathode rays are deflected toward the North Pole in a magnetic field.
- Cathode rays e gas to ionized.
- Cathode rays turn wheel paddle placed in their parts.
- Cathode rays produce heat energy.
- Cathode rays have a high penetrating power.
- Cathode rays affect photographic plates.
- Cathode rays can penetrate through some metals.
Application of gas discharge tube
Fluorescent lamps and neon signs:
The principle of the gas discharge tube is used fluorescent tubes for lighting and display of signs.
A fluorescent tube contains mercury vapour or sodium vapour which glow at low pressure when cathode rays pass through the vapour. The cathode rays produced inside the tube cause emission of ultraviolet rays when they bombard the vaporised atoms of mercury. The inside of the fluorescent lamp is coated with phosphor so that it gives off visible light when the ultraviolet rays strike it. Fluorescent tubes are also used as display signs. Neon gas is commonly used because it gives a bright orange light. Fluorescent lamps are more efficient than filament lamps.
The hot cathode
The hot cathode is another way of producing free electrons apart from what happened in a discharge tube when electrons are produced from a cold cathode. Whenever metal is heated to a sufficiently high temperature, electrons are emitted from the surface of the metal.
Thermionic emission
Thermionic emission is the process whereby electrons are emitted from a metal surface.
A diagram to explain thermionic emission:
Explanation of the operations
When the cathode or hot filament is heated to a high temperature by a voltage source across its terminal, the filament emits electrons by thermionic emission. Extra energy is given to the free electrons at the metal surface. The extra energy enables the electrons to break away from the metal surface and exist as free elections. The electrons accelerate to the positively biased anode and therefore produce a current which flows through the galvanometer.
Thermionic devices
These are devices that operate on the principle of thermionic emission. The devices:
- Diode valve
- X-ray tube
- Cathode ray tube
Explanation
Diode valve:
Diode value is a simple application of thermionic emission. It consists of an anode, filament made of tungsten wire and a cathode which surrounds the filament.
Diagram of the valve:
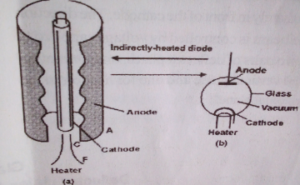
Operations of a diode valve
When the filament is heated to a high temperature by a voltage source across its terminal, the filament emits electrons by thermionic emission. The electrons accelerate to the positively biased anode and therefore produce a current which flows at the anode. The current can be detected by a galvanometer.
Diode valve characteristics
The diode value characteristics are used to show and explain the action of the diode valve.
Diode valve characteristic circuit:
Operations of the circuit
When the filament is heated to a high temperature by a voltage source across its terminal, the filament emits electrons by thermionic emission. The electrons accelerate to the positively biased anode and therefore produce anode current which flows at the anode. The current can be detected by a galvanometer. A voltmeter is connected across the anode and the cathode to know the anode voltage Va that produced the anode current Is. Different anode voltage Va is applied to obtain different anode current Ia. Each of the anode voltages Va is plotted its corresponding anode current Ia to obtain the diode valve characteristics.
Diode valve characteristics curve
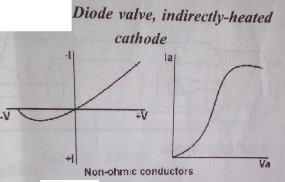
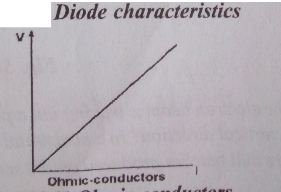
Explanation of the curve
The diode characteristics curve shows the behaviour of a diode valve. The cure shows that diode current Ia is not directly proportional to diode voltage Va. Initially, as the anode voltage Va is increased, the diode current Ia increases until it reaches its maximum value. But with further increase in anode voltage, the anode current remained constant or decreases. This point of the maximum value of Ia is called the saturation point. The anode current this point is called saturation current. For this reason, a diode valve does not obey ohms and it is called a non-ohmic conductor or substance. In a diode valve, current flow only in one direction.
Similarities between thermionic emission and photoelectric emission:
- Both of them are a surface phenomenon.
- Electrons are released in both cases.
- Energy is released in both cases.
- Energy transformation takes place in both cases.
- Increase in temperate is involved in both cases.
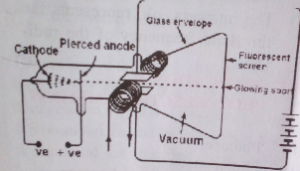
Cathode ray oscilloscope:
Cathode ray oscilloscope is the application of thermionic emission. It is a vacuum tube that contains an electron gun at one end and a fluorescent screen at the other end. Between these two are two pairs of reflectors that are used to control the electron unto the screen.
Diagram of cathode rays oscilloscope:
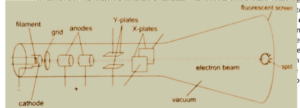
Explanation of operation of cathode rays oscilloscope
The heated filament heats the cathode and causes it to produces a stream of electrons by thermionic emission, which travels to the screen. The positively biased anodes accelerate the electron unto the screen. The X-plates are used to move the electron beam left and right while the y-plates move the electron beam up and down. The grid is used to regulate the amount of electrons that gravel to the screen and cause the screen to. Cooling fin is attached to cool the temperature of the tube.
General evaluation
- State the uses of a cathode-ray oscilloscope.
Assignment
- Define a thermionic emission and briefly explain how it is produced.
In our next class, we will be talking about the Wave Nature of Matter. We hope you enjoyed the class.
Should you have any further question, feel free to ask in the comment section below and trust us to respond as soon as possible.
